Plain Vanilla
An exerpt from the book Fast Food Nation by Eric Schlosser originally appeared in the Atlantic Monthly under the title “Why McDonald’s Fries Taste so Good”. While the entire piece was eye-opening, one particular aspect of it sparked a memory of another article, this one on ingredient fraud, which was remarkable in its own right.
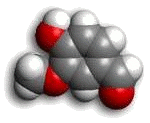
The Vanillin Molecule
As Scholsser points out in his piece, the specific molecules that make up particular food flavorings are exactly the same whether they are labeled “natural” or “artificial”. The only difference between them is the ultimate source of the molecule itself. Those derived from natural sources are “natural”; those that are man-made are “artificial” even though they are precisely the same:
“A natural flavor,” says Terry Acree, a professor of food science at Cornell University, “is a flavor that’s been derived with an out-of-date technology.” Natural flavors and artificial flavors sometimes contain exactly the same chemicals, produced through different methods. Amyl acetate, for example, provides the dominant note of banana flavor. When it is distilled from bananas with a solvent, amyl acetate is a natural flavor. When it is produced by mixing vinegar with amyl alcohol and adding sulfuric acid as a catalyst, amyl acetate is an artificial flavor. Either way it smells and tastes the same.
In Roald Hoffmann’s article “Fraudulant Molecules” in the Summer 1997 issue of American Scientist, he described the remarkable lengths to which some counterfeiters will go in order to secure the “natural” label. Quoting from an article in New Scientist:
[The] French food industry would need ten times as many vanilla pods as it actually imports to account for all the “‘natural” vanilla that companies claim is in milk products alone.
Clearly artificial vanilla was being fraudulently sold as natural vanilla; since the natural variety commands 200 times the price, it’s easy to understand the motivation behind the fraud. As there is no difference in the vanillin (the key molecule in vanilla) at the molecular level, methods of detection that exploit anomalies in constituent atomic isotopes have been devised. Of course, the counterfeiters responded with ever-more esoteric techniques to avoid detection.
- The first method involved detecting the ratio of naturally occurring radioactive carbon, 14C, which is continuously created in the upper atmosphere and absorbed by living material. Non-living material sees its level of 14C decay to 13C with a half-life of 5,730 years. (This is the process exploited by radio-carbon dating). The forgers responded by loading extra 14C to their petroleum-based precursors.
- Scientists then turned to the subtle differences in the ration of 13C and 12C which is it slightly lowered by the process of photosynthesis occurring in the vanilla plant. Once again, the forgers responded by applying appropriate amounts of 13C.
- Since it turned out to be most economical of add the additional 13C at a particular location on the vanillin molecule, its overabundance at that location could be detected using a technique called SNIF-NMR, that is, site-specific natural isotope fractionation by nuclear magnetic resonance.
At some level this has become almost comical. Humans cannot detect any difference in taste, and the split between the labels “natural” and “artificial” has become almost as artificial as the “fake” molecules themselves.


Concerning Plain vanilla what is its costs ? what is the means of delivery to your place if your are interested in buying it .We are situated in kenya .
» Posted by John Kagondo on May 15, 2007 05:22 AM